
CO 2 has one carbon atom and two oxygen atoms.Most periodic tables provide additional data (such as atomic mass) in a box that contains each element’s symbol. A modern version is shown in Figure 3.6.1 3.6. Oxygen (O) has an atomic mass of about 16.00 amu. The periodic table is one of the cornerstones of chemistry because it organizes all the known elements on the basis of their chemical properties.Carbon (C) has an atomic mass of about 12.01 amu.Let's calculate the molar mass of carbon dioxide (CO 2): Add them together: add the results from step 3 to get the total molar mass of the compound. 
Calculate molar mass of each element: multiply the atomic mass of each element by the number of atoms of that element in the compound.The atomic mass is usually found on the periodic table and is given in atomic mass units (amu). Find atomic masses: look up the atomic masses of each element present in the compound.The molar mass is used to convert grams of a substance to moles and is used often in chemistry. For example, water is H 2O, meaning it contains two hydrogen atoms and one oxygen atom. The latest release of the Periodic Table (dated ) includes the most recent abridged standard atomic weight values released by the IUPAC Commission on Isotopic Abundances and Atomic Weights (), compiled as part of the 2021 Table of Standard Atomic Weights 2021. The mass of a mole of substance is called the molar mass of that substance. This property is equivalent to what is today. (Figure 3.4.2 ), which he obtained by dividing the atomic mass (molar mass) in grams per mole by the density of the element in grams per cubic centimeter. Identify the compound: write down the chemical formula of the compound. The periodic table achieved its modern form through the work of the German chemist Julius Lothar Meyer (18301895) and the Russian.One mole contains exactly 6.022 ×10 23 particles (Avogadro's number) 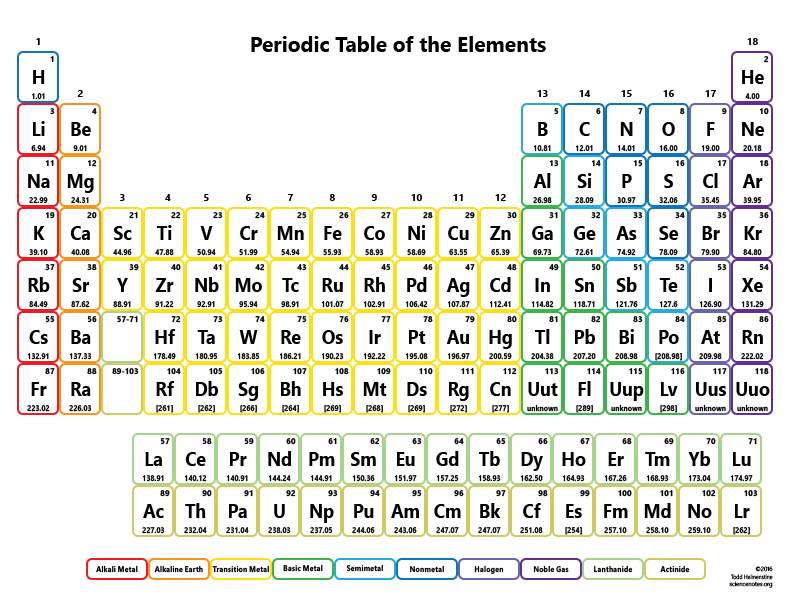
The molar mass of water is the mass of one mole of H 2 O.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
